
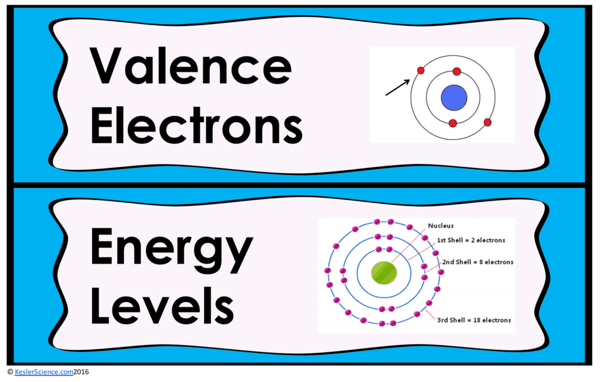
The teacher will help to clear any misconceptions about characteristics of organisms. A major misconception is that students do not understand prefixes, which may help them decode these difficult words.
Estimated Class Time for the Engagement: 20-30 minutes
EXPLORATION
This student-centered station lab is set up so students can begin to explore characteristics of organisms. Four of the stations are considered input stations where students are learning new information about cell theory and four of the stations are output stations where students will be demonstrating their mastery of the input stations. Each of the stations is differentiated to challenge students using a different learning style. You can read more about how I set up the station labs here.
EXPLORE IT!
Students will be working in pairs to better understand the characteristics of organisms. Students will be using images, vocabulary, and brief descriptions to match up. After the students have read up using information from other stations, the picture cards will be beneficial to make the concepts more concrete for students to understand. Students will record their observations on their lab sheet.
WATCH IT!
At this station, students will be watching a short video explaining asexual reproduction and how organisms benefit and suffer from it. Students will then answer some questions relating to the video and record their answers on their lab station sheet. For example: How many parents are involved in sexual reproduction? How many parents are involved in asexual reproduction? What percentage of the DNA is passed to the offspring in asexual reproduction? List 3 methods of asexual reproduction.
RESEARCH IT!
The research station will allow students to explore a web page that explains characteristics for the 6 Kingdoms. Students will be instructed to complete a few tasks and record answers on their lab sheets.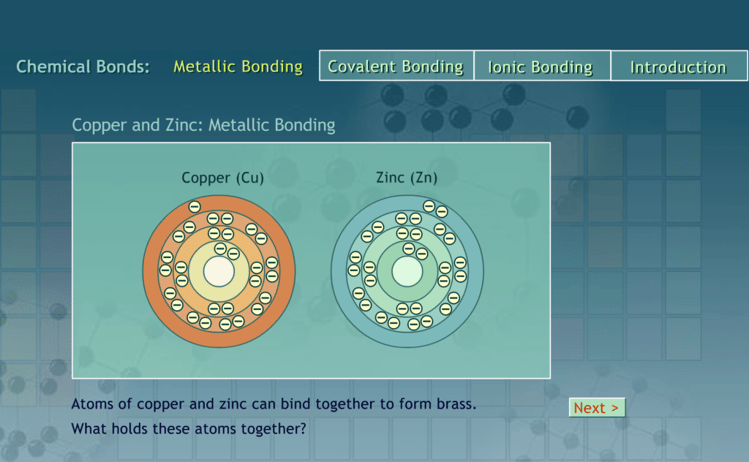
READ IT!
This station will provide students with a one page reading about the differences between autotrophs and heterotrophs. Students will read information about how autotrophs create their own food, heterotrophs have to find food, and the process of photosynthesis. There are 4 follow-up questions that the students will answer to show reading comprehension of the subject.
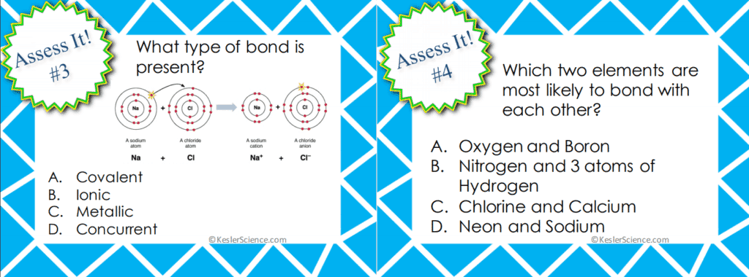
ASSESS IT!
The assess it station is where students will go to prove mastery over the concepts they learned in the lab. The questions are set up in a standardized format with multiple choice answers. Some questions will ask students: Which are the correct characteristics for bacteria? What is true about asexual reproduction? Which organism is autotrophic? An animal from the kingdom Animalia likely has which of the following characteristics?
WRITE IT!
Students who can answer open-ended questions about the lab truly understand the concepts that are being taught. At this station the students will be answering three task cards like: Describe the difference between sexual and asexual reproduction. Describe the difference between an autotroph and a heterotroph. Describe the difference between prokaryotic and eukaryotic cells.
ILLUSTRATE IT!
Your visual students will love this station. Students will draw 3 living things. Students will then have to think about the characteristic pairs and label their drawings with the correct selection of the 4 pairs of characteristics of organisms.

ORGANIZE IT!
The organize it station allows your students to match the characteristics of organisms with pictures of organisms provided. This station takes all information gathered from the other stations and challenges that students to recall the meanings of each of the characteristic descriptions and have them correctly identify which ones go with the pictures. Once students have completed their organization, the teacher will come and check their understanding.
Estimated Class Time for the Exploration: 1-2, 45 minute class periods
EXPLANATION
The explanation activities will become much more engaging for the class once they have completed the exploration station lab. During the explanation piece, the teacher will be clearing up any misconceptions about characteristics of organisms with an interactive PowerPoint, anchor charts, and interactive notebook activities.
The characteristics of organisms lesson includes a PowerPoint with activities scattered throughout to keep the students engaged.
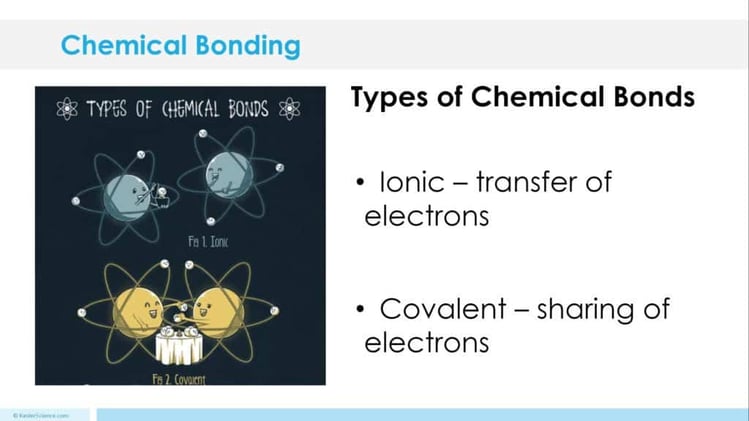
The students will also be interacting with their journals using INB templates for characteristics of organisms. Each INB activity is designed to help students compartmentalize information for a greater understanding of the concept. The characteristics of organisms INB templates allow students to focus their notes on understanding the description of each characteristic and how it stacks up next to its opposite characteristic description.

Estimated Class Time for the Exploration: 2-3, 45 minute class periods
ELABORATION
The elaboration section of the 5E method of instruction is intended to give students choice on how they can prove mastery of the concept. When students are given choice the ‘buy-in’ is much greater than when the teacher tells them the project they will have to create. The elaboration project will allow students to create either an infographic, pamphlet, diorama, computer presentation, or even a card game that helps to teach about the characteristics of organisms. Estimated Class Time for the Elaboration: 2-3, 45 minute class periods (can also be used as an at-home project)
Estimated Class Time for the Elaboration: 2-3, 45 minute class periods (can also be used as an at-home project)
EVALUATION
The final piece of the 5E model is to evaluate student comprehension. Included in every 5E lesson is a homework assignment, assessment, and modified assessment. Research has shown that homework needs to be meaningful and applicable to real-world activities in order to be effective. When possible, I like to give open-ended assessments to truly gauge the student’s comprehension.
Estimated Class Time for the Elaboration: 1, 45 minute class period
DOWNLOAD THE FULL LESSON NOW
The full lesson is available for download from my TpT store. Save yourself a ton of time and grab it now.






