
 The teacher will help to clear any misconceptions about counting atoms and elements. A couple major misconceptions are students do not understand the multiplication process using a coefficient. Also, students confuse the number of atoms with the number of elements.
The teacher will help to clear any misconceptions about counting atoms and elements. A couple major misconceptions are students do not understand the multiplication process using a coefficient. Also, students confuse the number of atoms with the number of elements.
Estimated Class Time for the Engagement: 20-30 minutes
EXPLORATION
This student-centered station lab is set up so students can begin to explore counting atoms and elements. Four of the stations are considered input stations where students are learning new information about counting atoms and elements, and four of the stations are output stations where students will be demonstrating their mastery of the input stations. Each of the stations is differentiated to challenge students using a different learning style. You can read more about how I set up the station labs here.
EXPLORE IT!
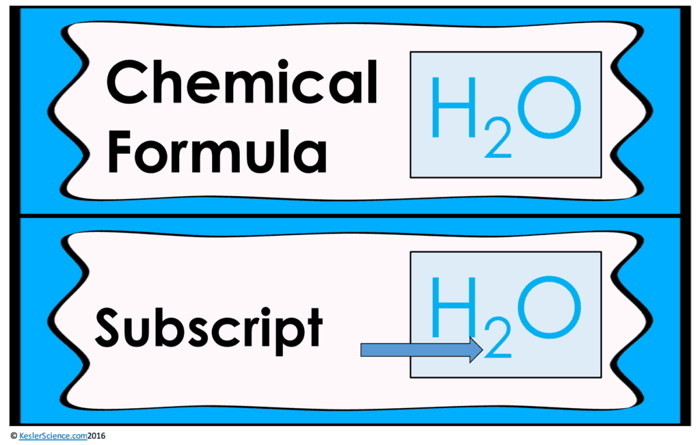
Students will be working in pairs to learn how to count the number of elements and atoms in three different compounds. Students will have to follow the directions on the task cards and will learn how coefficients change the number of atoms but not the number of elements. On certain cards, students are instructed to count the number of atoms of elements, and the total number of atoms in the compounds they are looking at.

WATCH IT!
At this station, students will be watching a three-minute video explaining how to count atoms in chemical formulas. Students will then answer questions relating to the video and record their answers on their lab station sheet. For example, what does a coefficient represent in a chemical formula? How do you use the coefficient to calculate the number of atoms in each molecule? How many total atoms are in the compound below?
RESEARCH IT!
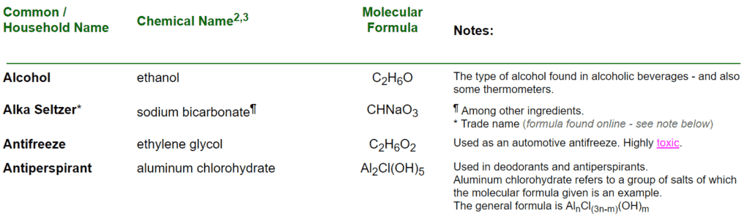
The research station will allow students to discover the compounds in common household items. Students will look up compounds for items such as Bleach, Chalk, Tylenol, Battery Acid, and even Antiperspirant. Once students discover the formulas, they will need to calculate the total number of atoms and the total number of elements in each compound.
READ IT!
This station will provide students with a one page reading about molecules. In the reading, students will discover how when elements bond together, they form molecules. The reading will take students from as far back as the formation of the solar system, to how today scientists are creating new molecules.There are 4 follow-up questions that the students will answer to show reading comprehension of the subject.
ASSESS IT!
The assess it station is where students will go to prove mastery over the concepts they learned in the lab. The questions are set up in a standardized format with multiple choice answers. Some questions include: How many elements are present in the compound below? How many total atoms are present in the compound below? How many atoms of Hydrogen are present in the compound below?
WRITE IT!
Students who can answer open-ended questions about the lab truly understand the concepts that are being taught. At this station, the students will be answering three task cards: Explain how to count the number of elements in a compound. Explain how to count the number of atoms in a molecule that has parentheses () around it. Explain how the total number of atoms is different from the total number of elements in a compound.
ILLUSTRATE IT!
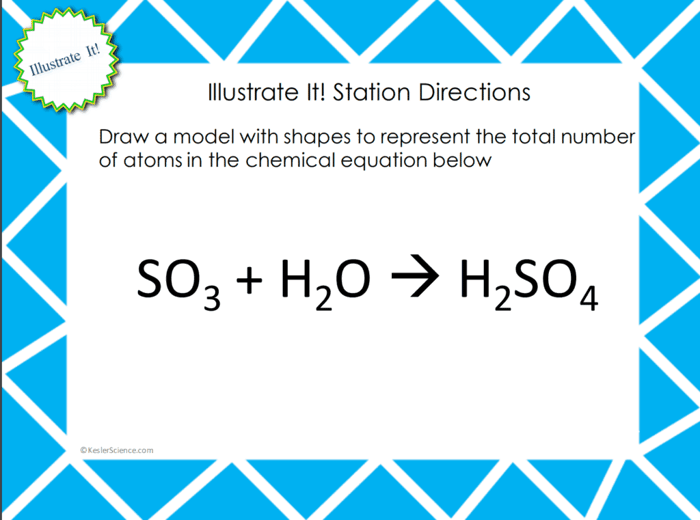
Your visual students will love this station. Students will draw a model of a chemical equation using different shapes to represent the different elements and the number of atoms for each element.
ORGANIZE IT!
The organize it station allows your students to place cards under the correct compound. Cards refer to the number of elements and the number of atoms that go with the correct compound.
Estimated Class Time for the Exploration: 1-2, 45 minute class periods
EXPLANATION
The explanation activities will become much more engaging for the class once they have completed the exploration station lab. During the explanation piece, the teacher will be clearing up any misconceptions with an interactive PowerPoint, anchor charts, and interactive notebook activities. The counting atoms and elements lesson includes a PowerPoint with activities scattered throughout to keep the students engaged.

The students will also be interacting with their journals using INB templates for counting atoms and elements. Each INB activity is designed to help students compartmentalize information for a greater understanding of the concept. The counting atoms and elements INB template allows students to focus their notes on learning how to count the number of atoms and elements in compounds. The INB templates also come with a number of practice compounds that allow students to master the concept.

Estimated Class Time for the Exploration: 2-3, 45 minute class periods
ELABORATION
The elaboration section of the 5E method of instruction is intended to give students choice on how they can prove mastery of the concept. When students are given choice the ‘buy-in’ is much greater than when the teacher tells them the project they will have to create. The elaboration project will allow students to create a presentation to teach about counting atoms and elements. Estimated Class Time for the Elaboration: 2-3, 45 minute class periods (can also be used as an at-home project)
Estimated Class Time for the Elaboration: 2-3, 45 minute class periods (can also be used as an at-home project)
EVALUATION
The final piece of the 5E model is to evaluate student comprehension. Included in every 5E lesson is a homework assignment, assessment, and modified assessment. Research has shown that homework needs to be meaningful and applicable to real-world activities in order to be effective. When possible, I like to give open-ended assessments to truly gauge the student’s comprehension.
Estimated Class Time for the Elaboration: 1, 45 minute class period
DOWNLOAD THE FULL LESSON NOW
The full lesson is available for download from my TpT store. Save yourself a ton of time and grab it now.





